To know about heat transfer first we must know about
energy. Energy is the ability to do work or produce power to do work . Energy can neither be created nor be destroyed but it can
change from one from to another. Energy can exist in various forms like
mechanical energy, chemical energy, electrical energy, nuclear energy, thermal
energy etc. Human takes energy from food and do work, an electric motor produce
work by electric energy.
Where,
energy. Energy is the ability to do work or produce power to do work . Energy can neither be created nor be destroyed but it can
change from one from to another. Energy can exist in various forms like
mechanical energy, chemical energy, electrical energy, nuclear energy, thermal
energy etc. Human takes energy from food and do work, an electric motor produce
work by electric energy.
Thermal energy is also called heat energy that can produce
work. If two body are at different temperature than heat is transfer from
higher temperature body to lower temperature body. So temperature is driving
force of heat.
work. If two body are at different temperature than heat is transfer from
higher temperature body to lower temperature body. So temperature is driving
force of heat.
The branch of science which deals with the transfer of energy
in form of heat is called heat transfer.
in form of heat is called heat transfer.
By the use of thermodynamic alone we can determine the total
heat transfer from a system for given change in its temperature.
heat transfer from a system for given change in its temperature.
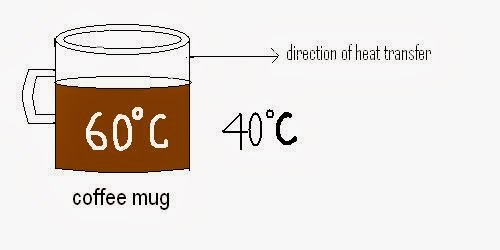
For example: If we interested in determining that how much heat is transferred by a coffee mug to cool down from 600C to 400C. Then this can be find out by using the branch thermodynamic.
Through thermodynamic by using the formula given below we can calculate the amount of heat transferred by the coffee mug to its surroundings.
Q = mCp (t2 – t1)
Where,
t2 = Final temperature of coffee mug,
t1 = Initial temperature of coffee mug,
Q = Total heat transfer,
m = Mass of coffee,
Cp = Specific heat of coffee
But this analysis cannot tell us that in how much time the
mug of coffee cool down. So we need heat transfer that tell us about the rate of heat
transfer and we can calculate the time taken by the coffee mug to cool down.
mug of coffee cool down. So we need heat transfer that tell us about the rate of heat
transfer and we can calculate the time taken by the coffee mug to cool down.
T = Q / q
Where,
T = Time taken by the coffee mug to cool down
Q = Total heat transfer,
q = Heat transfer rate which is determined by heat transfer
analysis.
analysis.
Heat transfer rate is basically depends upon the medium existing between the two body. It is a wide area of science to study.

.bmp)







