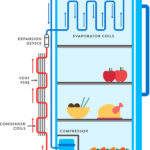
Adiabatic process is a process in which there is no exchange of heat takes place from the working substance to the surrounding during its expansion or compression. This is possible when the working system is made thermally insulated, that is no heat can leave or enters it during the process. So we can say that during adiabatic or isentropic process.
1. Heat does not leaves or enters the working substance (usually a gas).
2. The temperature of the gas changes as the work is done on the cost of internal energy.
3. The work done is equal to the change in internal energy.
Types
This process may be categorized as reversible or irreversible.
- Reversible or frictionless adiabatic process is called as isentropic process (in which entropy remains
constant) - When the friction is considered in the process than the process is taken as irreversible adiabatic process in which the entropy does not remains constant (i.e. entropy increases)
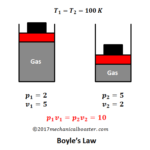
Let’s us considered m kg of air is heated adiabatically from initial state 1 to final state 2 and
The P-V diagram of the process is given below:
Let
p1, v1 and T1 = Pressure, volume and temperature at initial stage 1 and
p2, v2 and T2 = Pressure, volume and temperature at final state 2.
In the P-V diagram that is shown above, a gas in the piston cylinder assembly is heated adiabatically (i.e. not heat leaves or enters into the gas. To prevent the heat exchange from the system, the walls of the cylinder are made perfectly non-conductor. As the gas inside the cylinder is heated, the pressure, volume and temperature increases and it starts to expand.
The various relationship of the reversible adiabatic process are given below:




.bmp)